Drug Import License Re-registration in India - Form 40 & Form 41

Are you a foreign manufacturer who wishes to import or renew their import license for sale in the Indian market? Your one-stop shop for successfully and quickly handling the crucial Form 41 licensing process is CliniExperts. Get in touch with us right now to arrange a free consultation and go over how we can assist you with getting your license.
Re-registration – Overview
- CliniExperts will handle the submission of Form 40 and all necessary documents if a foreign company wishes to enter the Indian market and does not have a local presence.
- CliniExperts carefully reviews all documents, performs a GAP Analysis to address any deficiencies, and promptly communicates any issues for a seamless application process.
- We manage the final review of your application and coordinate the payment of government fees for efficient financial handling.
- We ensure all submissions comply with the Drugs and Cosmetics Act, including the required undertakings in Schedule D-I and D-II.
- Our expertise lies in preparing and submitting Form 40 applications to the CDSCO, assisting with the renewal of Form 41 registration certificates for importing pharmaceuticals into India, and ensuring full compliance with CDSCO regulations.

Who Can Apply?
Renewal applications are only available to importers.
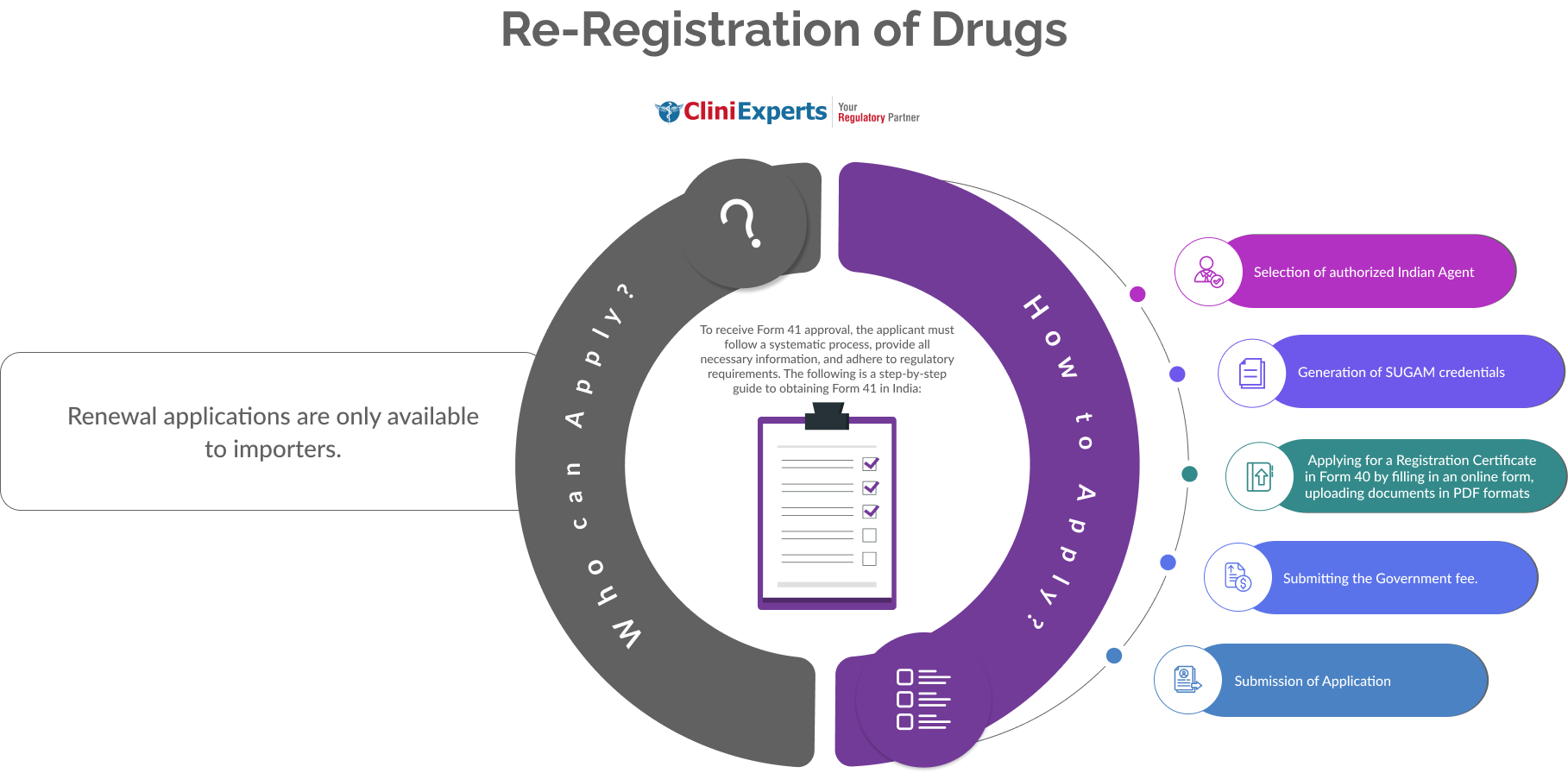
How To Apply?
The Applicant must follow the following process:
-

Selection of authorized Indian Agent or appoint CliniExperts as your Authorized agent in India.
-

Generation of SUGAM credentials
-

Applying for a Registration Certificate in Form 40 by filling in an online form, uploading documents in PDF formats
-

Submitting the Government fee.
-

Submission of Application

Validity
The registration certificate will be valid for three years. Post this, it should be renewed again by following the same process.
Fee Involved
The following fees and forms must be submitted. This includes undertakings required under Schedule D(I) for manufacturing facility registration and Schedule D(II) for pharmaceutical registration.- A registration cost of USD 10,000 (or the equivalent amount in Indian currency) for the manufacturing premises.
- A registration fee of USD 5,000 (or its equivalent in Indian currency) is required for one drug. An additional USD 5,000 (or its equivalent in Indian currency) is necessary for each additional drug, as long as the production location remains the same. Fees must be paid using a Challan at the Bank of Baroda, located at Kasturba Gandhi Marg, New Delhi 110 001, or at any other bank notified by the authorities.
Important Documents

- Wholesale license and/or manufacturing license.
- Drug master file
- Site master file
- Free sale certificate/Certificate of Pharmaceutical Product
- Good Manufacturing Practice Certificate
- Power of attorney
Timeline to get
Form 41
from Central Drugs Standard Control Organisation
9
MonthsEssential Tips
- At the time of application, stability data spanning at least six months is available.
- The supporting paperwork for an application needs to be apostilled or notarized.
- The authorized product's indication, strength, dosage form, and administration method shouldn't be altered.
- The applicant's or manufacturer's submitted documents must contain accurate name and address information.
Expert Advise
According to the experts:
The company needs to confirm that the manufacturing site for which they are applying has never before received a registration certificate.
The manufacturer is responsible for verifying that all regulatory documents are still valid. The product samples that are sent to the government laboratory for testing ought to be in adequate numbers and well within the product's shelf life.
Why Choose CliniExperts?
By partnering with CliniExperts, manufacturers benefit from a streamlined, expert-led approach that minimizes the complexity of regulatory compliance, enhances the accuracy of your application, and accelerates your entry into the Indian market.

Expert Guidance
CliniExperts assist with the registration process with regulatory bodies and ensure compliance with all regulatory requirements, including the timely submission of necessary documents.

Thorough Document Review
CliniExperts conduct a meticulous review of all documents, perform a GAP Analysis to identify and address any deficiencies, and communicate any issues promptly to ensure a smooth application process.
Final Review and Fee Management
CliniExperts oversee the final review of your application and coordinate the payment of government fees, ensuring that all financial aspects are handled efficiently.

Timely Follow up
CliniExperts will maintain timely follow-ups with the relevant Government bodies to keep everything on track.

Regulatory Compliance
CliniExperts ensure that all submissions are in strict adherence to the regulatory guidelines.
Frequently Asked Questions
Should someone obtain a registration certificate for Drugs that are going to be transported from India to other nations but will not be sold or distributed there?
No, in situations where only transportation is required, a registration certificate is not required.

How long will the registration certificate be valid if the address of the manufacturing site using Form 41 and the firm's constitution both change?

What are the main obstacles to the registration of drugs in India?
The complicated regulatory environment, strict documentation requirements, controlling expenses and time restrictions, guaranteeing compliance with Indian biotech product rules, and accurate interpretation of guidelines are some of the major obstacles in the Drugs medication registration process.

