COVID-19 Home Testing Using Rapid Antigen Tests (RATs)

Rapid Antigen tests (RATs) are commonly used to diagnose respiratory pathogens, including influenza virus and SARS-CoV-2 virus. These tests have come to practical use in the diagnosis of the infection in the Covid-19 pandemic. People with suspected COVID-19 need to know immediately if they are infected to take the necessary precautions like home isolation, taking treatment and informing close contacts. Home test kits can immensely expedite this entire process.
Rapid Antigen Home Tests:
People can now do rapid antigen tests at home, that are easily accessible but have limited condition to their usage. This test is advised to be done only in those people who show symptoms related to the infection and people who are in close contact with the positively tested patients. Random testing is not advisable.
The Rapid antigen home test is done with the help of mobile apps, which are available for installation on the Google Play Store and Apple Store. The test should be conducted by carefully following the steps given in the user manual. The pictorial and video link of the user manual is shown on the test kit.
After complete registration, the mobile app provides a guide that makes it easier to procure the test results for the patient. After completing the test procedure, the user is advised to click a picture of the test strip with the same mobile phone with the preinstalled app. All the data that appears in the mobile phone is compiled and eventually stored in a secure server connected with the ICMR COVID-19 testing portal without any risk of disclosure in patient’s data confidentiality.[1]
Interpretation of the Test Results:
- People who are tested positive are considered true positives, and there is no need to test again. Everyone who is tested positive should maintain home isolation and care as per the ICMR & Health Ministry protocols.
- People who are tested negative by the RAT should get tested by RTPCR. The negative test results of RAT can be due to misinterpretation in the testing of favourable products with low viral load, and all RAT negative individuals may be treated as suspect of COVID-19 until they have received confirmation of the RTPCR test result.
- The test results should be interpreted and confirmed according to the protocol given in the user manual.
- After completing the test, people should follow instructions in the disposal of the test kit, swab and other materials.
Approved kit for Home Testing:
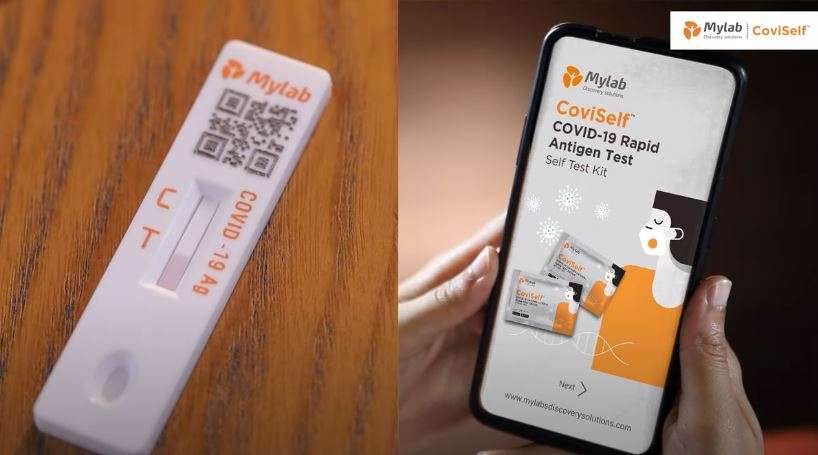
| Sr.no | Name of Company | Name of Kit | Sample Collected | Guidance Documents |
| 1. | Mylab Discovery Solutions Ltd. Pune (Maharashtra), India | CoviSelf (PathoCatch) COVID-19 OTC Antigen LF Device | Nasal Swab | Link for Instructions for use in Hindi and English-https://coviself.com/ifu/ Video link for demonstration video in Hindi and English- https://coviself.com/video/ |
Summary:
- The introduction of Rapid Antigen Home tests allows access to testing for more people who show COVID-19 symptoms and provides faster results to take action quickly to reduce the spread of infection.
- The Rapid Antigen Home tests come in a kit with a user manual, pictures and video links, which provide a guide to follow the steps to be taken to do the test.
- The test requires installing and registering mobile apps, which helps to proceed with the test results.
- People must carefully interpret test results according to the manufacturer’s instructions given in the test kit.
References:
- Advisory for COVID-19 Home testing using Rapid Antigen Tests (RATs), Indian Council of Medical Research [Internet] Updated: May 19, 2021, Accessed on May 25, 2021
- ICMR approves Mylab’s Covid-19 self-testing kit CoviSelf, issues home testing advisory, Healthworld.com [Internet] Available at: https://health.economictimes.indiatimes.com/news/diagnostics/icmr-approves-mylabs-covid-19-self-testing-kit-coviself-issues-home-testing-advisory/82780394 Accessed on May 25, 2021
Recent Posts
Role & Responsibilities of Authorized Agent for Drugs in India

The role of authorized agent responsibilities is critical for foreign Pharmaceutical companies seeking to register, Import, and market Drugs in India while ensuring full regulatory compliance wi..
How to Get a CDSCO Import License in India: A Step-by-Step Guide for Foreign Medical Device Companies

In India given its large population and ample growth opportunities, remains one of the most attractive markets for global Medical Devices company. But entering Indian market is not so easy as it not o..
Pharmacovigilance Regulations For Imported Drugs In India

Pharmacovigilance ensures that Drugs are continuously monitored for safety, quality, and efficacy through structured reporting systems and regulatory oversight mechanisms post-approval. In India, the ..
Need Help?
Submit your EnquiryOffice Locations
India
- Delhi
- Bangalore
Global
- USA
- Singapore
Call us on
Sales: +91 7672005050
Reception: +91-11-45214546
Timings
9 am to 6 pm (Monday to Friday)


