Table of Contents
How to Get a CDSCO Import License in India: A Step-by-Step Guide for Foreign Medical Device Companies

In India given its large population and ample growth opportunities, remains one of the most attractive markets for global Medical Devices company. But entering Indian market is not so easy as it not only involves commercial decision— but also involves defining correct regulatory pathway The Central Drugs Standard Control Organisation (CDSCO) regulates the Medical Devices in India under the Drugs and Cosmetics Act, 1940 and the Medical Devices Rules, 2017, with a risk-based classification and license covering devices from Class A to D.
For overseas manufacturers, one rule matters immediately: foreign companies do not typically enter the Indian Medical Device market by filing the application directly on their own. The import route generally requires working through an Indian authorized agent, and for existing devices the application pathway is commonly Form MD-14 followed by grant in Form MD-15.
Start with the right device classification
Before preparing for any application, a manufacturer needs to identify the device’s regulatory class. India uses a four-level risk framework:
Table1:
| Class | Risk Level |
|---|---|
| A | Low risk |
| B | Low–moderate risk |
| C | Moderate–high risk |
| D | High risk |
This is not a minor administrative detail. Classification affects the licensing pathway, documentation expectations, and the level of scrutiny your submission may receive. The official CDSCO Medical Device section also reflects a structured risk-based system under the Medical Devices Rules, 2017.
Appoint an Indian authorized agent
A foreign manufacturer without its own eligible Indian establishment usually needs an authorized agent to support the import licensing process. CliniExperts’ importer page explains that overseas manufacturers seeking to market their Medical Devices in India must route the process through an authorized licensing agent, and that these representative supports import and compliance responsibilities in India. This appointment is also significant from a legal and long-term strategic perspective. The official CDSCO FAQ states that a Power of Attorney and an undertaking from the authorized agent are required for the import license process, and it further clarifies that a change in Indian agent requires a fresh import license. That means choosing the right agent upfront can help avoid expensive rework later.
Note for manufacturers – It is important for manufacturer to ensure that the authorized agent have valid MD 42 from CDSCO. This license authorizes the Indian Agent to import, sell, stock, exhibit, or distribute Medical Devices, including In-Vitro diagnostic devices, to retailers, wholesalers.
Prepare the correct import license pathway
For existing Medical Devices, the CliniExperts importer page identifies the standard route as MD-14 for the application and MD-15 for the import license grant.
For new Medical Devices, the pathway can be more complex. The same page describes a three-layer permission structure that may involve the following MD forms:.
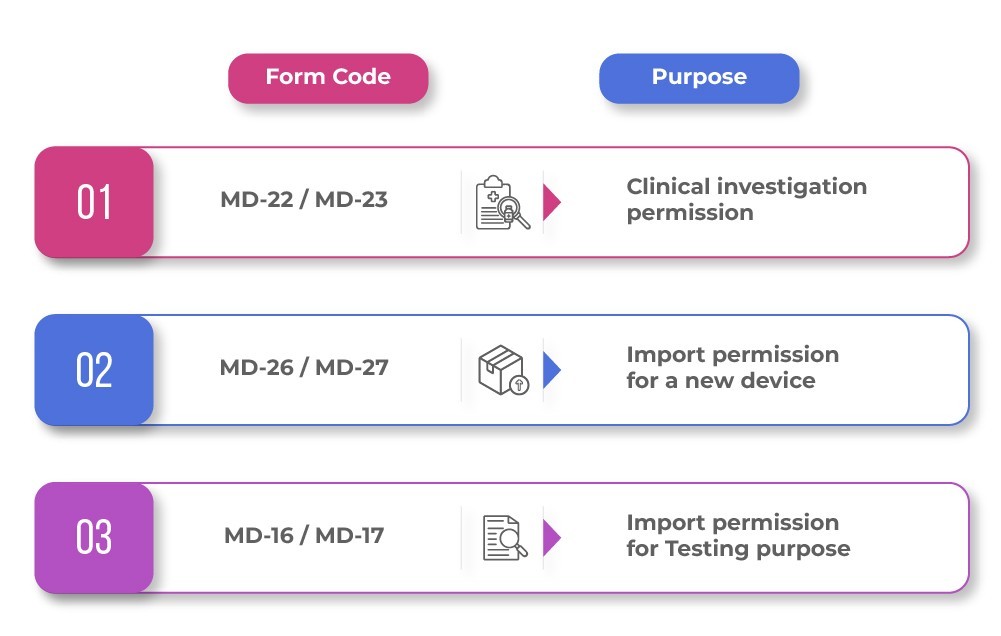
So, while some companies think “import license” means one standard form, the actual route depends on whether the product is already established also called predicate device or treated as a new device in India.
File through the SUGAM portal and manage follow-up carefully
India’s Medical Device licensing process is increasingly digital. CliniExperts note that registration and application workflows are handled through the SUGAM portal, while the official CDSCO online Medical Device portal functions as the digital interface for regulatory activity.
That means submission is only one part of the process. Application submission, Query management, document consistency, and timely follow-up all matter. Many delays happen not because companies do not know the form number, but because the submission file is incomplete, inconsistent, or poorly adapted to Indian expectations.
Common mistakes foreign manufacturers should avoid
The most common mistakes are predictable: choosing an agent too quickly, assuming classification is obvious, reusing global documents without adapting them for India, and failing to confirm whether the device is considered “new” in the Indian context. Even small errors can change timelines for device approval, trigger queries or re-filling the application.
Conclusion
For overseas Medical Device companies, India is a promising market, but their success depends on regulatory planning from day one. The most effective route is usually straightforward: classify the device correctly as per the rules, appoint the right authorized agent, fill out the correct MD Form, prepare the right set of documents, , and submit the application on SUGAM portal carefully. The companies that move fast are usually the ones that get these fundamentals right early.
Recent Posts
Role & Responsibilities of Authorized Agent for Drugs in India

The role of authorized agent responsibilities is critical for foreign Pharmaceutical companies seeking to register, Import, and market Drugs in India while ensuring full regulatory compliance wi..
Pharmacovigilance Regulations For Imported Drugs In India

Pharmacovigilance ensures that Drugs are continuously monitored for safety, quality, and efficacy through structured reporting systems and regulatory oversight mechanisms post-approval. In India, the ..
Need Help?
Submit your EnquiryOffice Locations
India
- Delhi
- Bangalore
Global
- USA
- Singapore
Call us on
Sales: +91 7672005050
Reception: +91-11-45214546
Timings
9 am to 6 pm (Monday to Friday)


