Permission for Clinical Trial (Phase 1, 2, 3) in India - CT-04 & CT-06

CliniExperts specializes in offering comprehensive regulatory solutions designed specifically for clinical trials. Get CT-06 approval in ease by choosing us and ensure prompt and efficient authorization for your clinical trial products.
Permission for Clinical Trial (Phase 1, 2, 3) – Overview

Who Can Apply?
Both manufacturers and importers are eligible to apply. The condition is that they must have an Indian entity with a manufacturing license in form 28/28A and a wholesale license in 20B/21B.
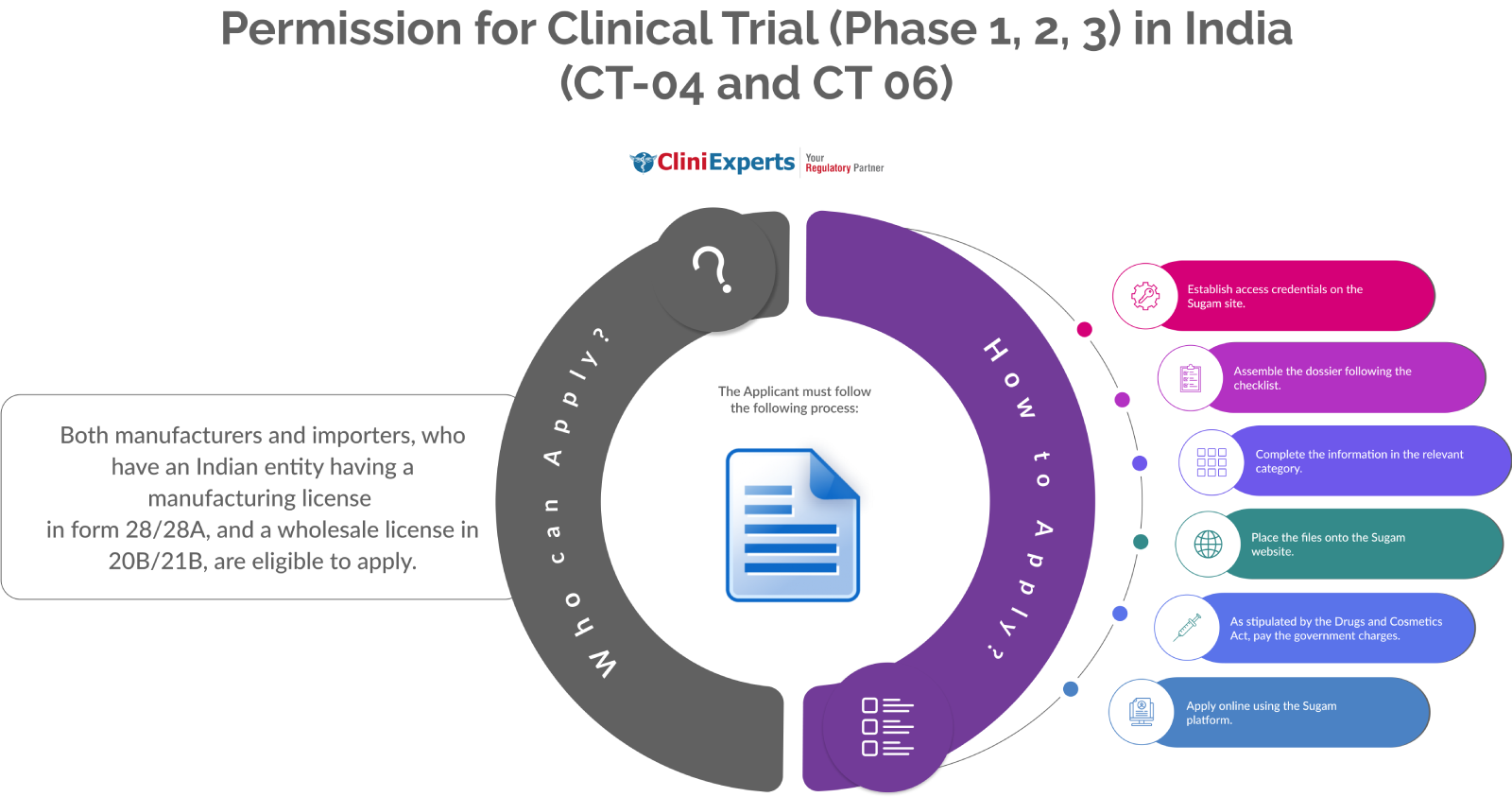
How To Apply?
The Applicant must follow the following process:
-

Establish access credentials on the Sugam site.
-

Assemble the dossier following the checklist.
-

Complete the information in the relevant category.
-
Place the files onto the Sugam website.
-

As stipulated by the Drugs and Cosmetics Act, pay the government charges.
-

Apply online using the Sugam platform.

Validity
The license is valid for 2 years.

Fee Involved
The government charges for the application process are as follows:
- Phase 1: 3 lacs
- Phase 2 and Phase 3: 2 lacs each
Important Documents

The documents mentioned below should be present during the documentation process:
- Chemistry manufacturing and control data
- Protocol for clinical trial
- Principle investigator undertaking
- Investigator brochure
- Informed consent form
- Case report form
- Investigational Medicinal Product Dossier
Timeline to get
CT-06
from Central Drugs Standard Control Organisation
90
DaysEssential Tips
The primary considerations to be made while preparing and submitting the license application are:
- The protocol we are submitting must be pre-approved by the European Commission Ethical committee.
- It is important to sign and stamp all documents with the consent of all parties involved.
- Applicants should note that 50% of the sites must be approved by the government. Also, it must be distributed geographically throughout India.
- The obstacles or issues that are most likely to arise and can be easily avoided by ensuring that all documents are in place, when filling out the form are:
- To choose their clinical trial sites carefully.
- ICF and CRF must comply with the NDCT Rule.
Expert Advise
According to the experts, clients should look out for the following during the application procedure:
Documents should follow the checklist.
Ethical Committee (EC) approval must be obtained for all clinical trial sites.
EC’s approval is required for the protocol.
Frequently Asked Questions
How to select the sites to perform clinical trials?
All trial sites must be distributed geographically throughout India and the applicant should note that 50% of sites must be government-approved.

Who should be a member of the EC Committee?
The following should be the members of the EC Committee:
- Chairperson/ Vice Chairperson (optional) Non-affiliated
- Member Secretary/ Alternate Member Secretary (optional) Affiliated
- Basic Medical Scientist(s) Affiliated/ non-affiliated
- Clinician(s) Affiliated/ non-affiliated
- Legal expert/s Affiliated/ non-affiliated
- Social scientist/ philosopher/ ethicist/theologian Affiliated/ non-affiliated
- Layperson(s) Non-affiliated

What is the government timeline to obtain Permission?
The license usually takes 90 days to obtain.

What is the government fee for permission?
The government charges are as follows:
- Phase 1: 3 lacs
- Phase 2 and Phase 3: 2 lacs each.

