Table of Contents
Reduced Approval Timelines and Enhanced Compliance Obligations under NDCT Amendment Rules 2026

The Ministry of Health and Family Welfare, through G.S.R. 46(E) dated 20 January 2026, has implemented critical amendments to the New Drugs and Clinical Trials (NDCT) Rules, 2019. A major aspect of this reform is the reduction and standardization of regulatory timelines for processing applications related to test licenses for new drugs, investigational new drugs (INDs), and unapproved active pharmaceutical ingredients (APIs). The updated timelines aim to facilitate faster research progression while preserving regulatory oversight.
These changes are designed to promote ease of doing research without diluting regulatory oversight, ensuring patient safety, data integrity, and accountability across the clinical trial lifecycle.
Background of NDCT Amendment Rules 2026
Under the NDCT Rules 2019, timelines—particularly for test license applications—were 90 working days and in practice often contributed to delays in initiating clinical trial material manufacturing. Stakeholders raised concerns during the draft amendment consultation in August 2025, leading the Central Government to reassess and revise these statutory durations.
The 2026 amendment formalizes 45 working days as the standard evaluation period, providing greater certainty and responsiveness within the regulatory framework.
Reduced Test License Approval Timeline India
A key highlight of the NDCT Amendment Rules 2026 is the clearly defined test license approval timeline India, replacing uncertainty with predictable regulatory outcomes.
Under the revised framework, CDSCO is required to process complete applications within a fixed statutory period.
- Rule 53 — Test License for New Drugs / Investigational New Drugs
The amendment substitutes all references to “ninety working days” with “forty-five working days.”
Thus, the Central Licensing Authority (CLA) must now review Form CT-10 applications for test licenses within 45 working days.
This applies to:
- Manufacturing for clinical trials
- Bioavailability (BA) or bioequivalence (BE) studies
- Manufacturing for examination, test, and analysis
- Rule 60 — Test License for Unapproved APIs or Formulations
Similarly, amendments to Rule 60 mandate a 45 working days decision period for:
- Form CT-14 (formulations)
- Form CT-15 (APIs)
All prior references to 90 days are replaced with 45 days.
This ensures harmonization between investigational product manufacturing and API/formulation-level test license approvals.
| Regulatory Area | Earlier Timeline | Updated Timeline (2026) |
| Test license for new drugs / INDs (Rule 53) | 90 working days | 45 working days |
| Test license for unapproved APIs / formulations (Rule 60) | 90 working days | 45 working days |
| Acknowledgment-based “Prior Intimation” | Not applicable | Immediate start upon acknowledgment |
| Appeal timelines | Unchanged | Unchanged, but clarified |
Enhanced CDSCO Test License Compliance Obligations
While timelines are reduced, CDSCO test license compliance obligations have been substantially strengthened under the amended rules.
Applicants must now demonstrate continuous compliance rather than one-time regulatory clearance.
This ensures that faster approvals do not compromise safety or regulatory rigor.
Key Compliance Requirements
- Adherence to GMP compliance NDCT Rules
- Timely record maintenance NDCT Rules
- Readiness for CDSCO inspection requirements
- Immediate reporting of deviations or violations
- Statutory Timeline Reduced from 90 to 45 Working Days
- Faster High-Risk Category Processing Despite Permission Route
- Timelines Updated in Multiple Sub-rules
- Timelines Apply Only to Permission Route, Not Prior Intimation
- Ensures Faster Initiation of Clinical Trial Material Manufacturing
GMP Compliance and Inspection Requirements
GMP compliance NDCT Rules have been reinforced, especially for facilities involved in manufacturing or testing investigational products.
CDSCO inspection requirements now extend beyond pre-approval stages to include routine and for-cause inspections.
Facilities must maintain updated documentation, validation records, and quality systems aligned with clinical trial standards.
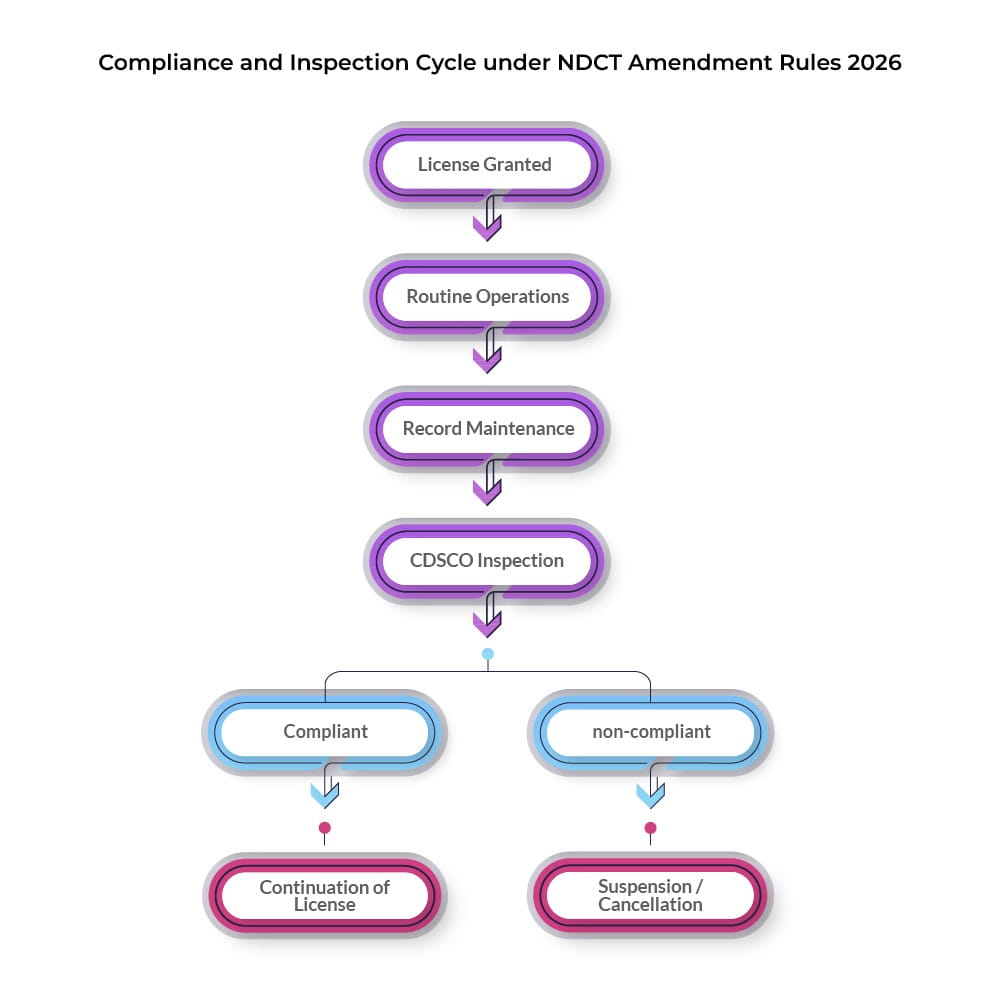
Test License Suspension and Cancellation Provisions
The NDCT Amendment Rules 2026 clearly define circumstances for test license suspension cancellation, enhancing regulatory transparency.
Non-compliance with GMP standards, falsification of records, or obstruction of inspections may trigger regulatory action.
License holders are provided with an opportunity to be heard before final orders are issued.
Appeal Provisions under Rules 53 to 60 NDCT
To balance enforcement powers, the amendment strengthens appeal provisions of NDCT Rules, ensuring procedural fairness.
Aggrieved parties may appeal for suspension or cancellation of orders within the prescribed timeframe.
These provisions reinforce trust in the regulatory system while maintaining strict oversight.
Regulatory Compliance Clinical Trials India: Practical Impact
For stakeholders involved in regulatory compliance clinical trials India, the amendments demand stronger internal governance systems.
Organizations must invest in compliance training, documentation systems, and audit preparedness.
However, the benefit lies in faster approvals and improved global credibility of Indian clinical research.
Conclusion and Key Takeaways
The NDCT Amendment Rules 2026 represent a balanced regulatory evolution, combining speed with responsibility.
Summary
- Defined 45 working days approval timeline improves predictability
- Compliance obligations under CDSCO are significantly enhanced
- GMP and inspection requirements are more rigorous
- Clear suspension, cancellation, and appeal mechanisms introduced
- Rules 53 to 60 NDCT strengthen regulatory enforcement
Overall, the NDCT Amendment Rules 2026 create a faster yet more accountable test licensing environment, supporting innovation while safeguarding compliance and patient safety in India’s clinical trial ecosystem.
Saurangi is a food regulatory expert with 8 years of experience. She shares her knowledge and insights on regulatory updates, food trends, best practices, and news. Follow her for expert insights and practical advice on all things for food regulatory
Saurangi Shah
CliniExperts Services Pvt. Ltd.
Recent Posts
Introduction of “Permission or Prior Intimation” Mechanism for Test Licenses under NDCT Rules, 2026

The NDCT Rules 2026 test license framework introduces a structured permission or prior intimation mechanism, streamlining regulatory oversight for investigational products, APIs, and clinical trial ma..
India-EU Free Trade Agreement (FTA): A Strategic Boost for India’s Pharma, Healthcare, and Food Sectors

The India-EU Free Trade Agreement (FTA) is a comprehensive trade framework aimed at liberalising bilateral trade and strengthening economic ties between India and the European Union. It enhances marke..
Need Help?
Submit your EnquiryOffice Locations
India
- Delhi
- Bangalore
Global
- USA
- Singapore
Call us on
Sales: +91 7672005050
Reception: +91-11-45214546
Timings
9 am to 6 pm (Monday to Friday)


