- Home
- Medical Devices Regulatory Services
![]()
Medical Devices Regulatory Services
Medical Devices Regulatory Services
Presently, the regulations for medical devices in India are still evolving. Only a select few products need a medical device permission to be launched. In the past few years, there have been three major announcements outlining the medical device products that need to be registered.
Up to 2005, only medical devices such as disposable hypodermic syringes, condoms, tubal rings, metered dose inhalers (MDIs), etc., had to be registered in India. Beginning March 1, 2006, items such as cardiac stents, catheters, intraocular lenses, bone cement,drug eluting stents, also must be registered for import, manufacture and sale. Further, on March 20, 2009, the Central Drugs Standard Control Organization (CDSCO) ratified that sterile medical equipments such as spinal needles, extension tubes, endotracheal tubes, and cardiac patches must also be registered.
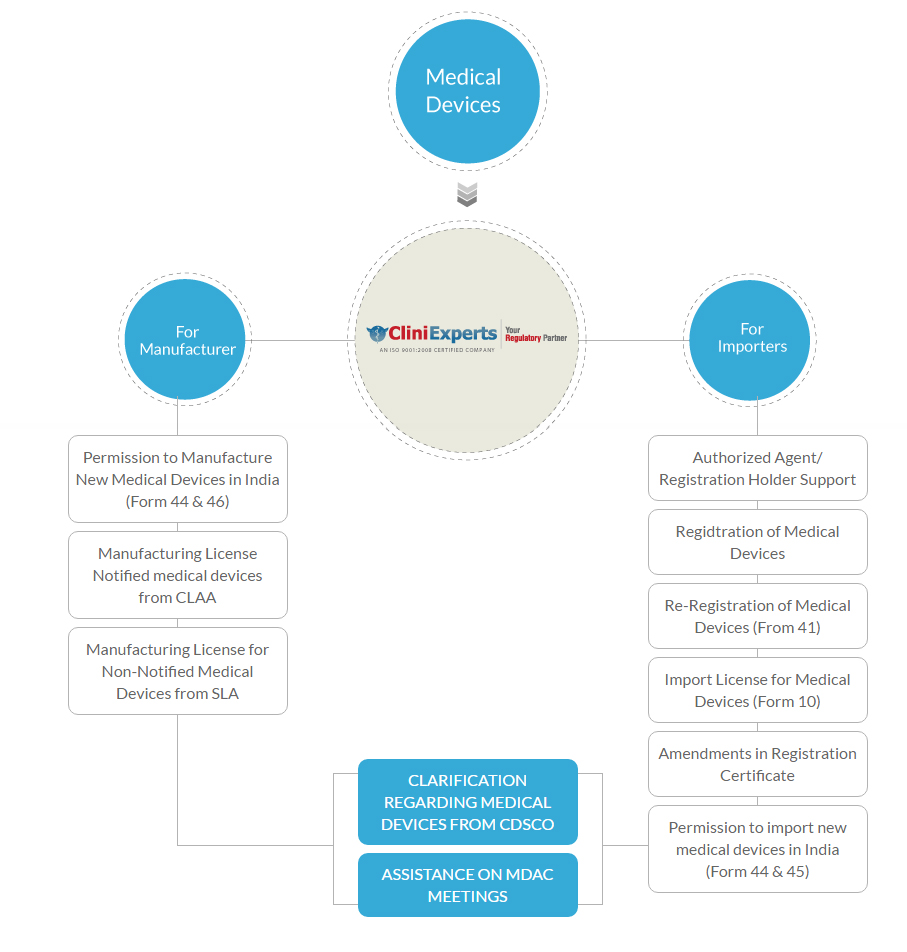
- For Manufacturer
- Permission to Manufacture New Medical Devices in India (Form 44 & 46)
- Manufacturing License Notified medical devices from CLAA
- Manufacturing License for Non-Notified Medical Devices from SLA
- For Importers
- Authorized Agent/ Registration Holder Support
- Registration of Medical Devices
- Re-Registration of Medical Devices (From 41)
- Import License for Medical Devices (Form 10)
- Amendments in Registration Certificate
- Permission to import new medical devices in India (Form 44 & 45)
For Importers
India is a huge and growing market for all healthcare products. Thus, an increasing number of foreign medical equipment companies are expected to venture and import their products. With this, even the regulatory situation on licenses and medical device registration in India is evolving continuously, and companies need to stay up-to-date in order to achieve success in India. With our assistance and up to date regulatory data base, our clients can plan and meticulously execute their projects in India.
Medical device regulation is still not completely defined in Indian regulations; hence, understanding the product and executing projects is a huge challenge .We at CliniExperts develop proper regulatory strategy and share it with our clients, which help build support for the strategy and ensure that CliniExperts held to its promise of executing the projects.
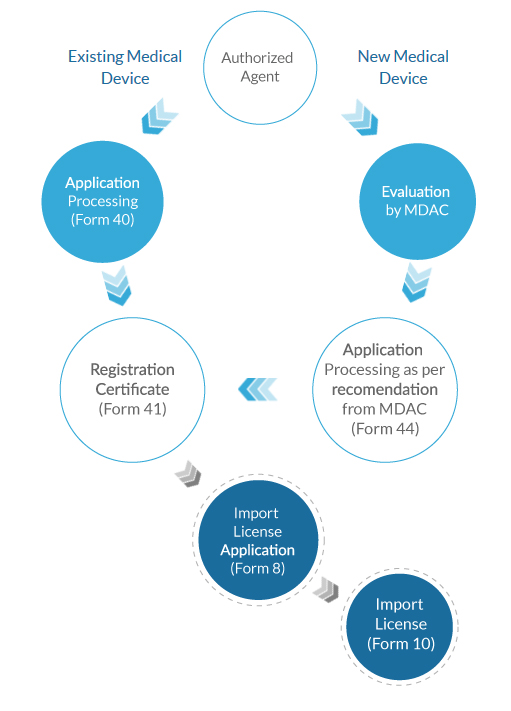
Registration Process
- Existing Medical
Device 
- New Medical
Device 





- Authorized agent/
Registration holder support - Registration of Medical Devices
(Form 40, Form 41) - Re-registration of
Medical Devices (Form 41) - Import License for
Medical Devices (Form 8, Form 10) - Amendments in Registration Certificate
- Marketing Authorization for Import New Medical Devices in India (Form 44 and 45)
Authorized agent/Registration holder support
Foreign manufacturers without any physical presence in India cannot import a medical equipment. A local office or Indian Local Agent with a valid Wholesale License must be present to enable the import of devices in India.
We at CliniExperts hold a valid Wholesale License (Form 20B and 21B) enabling us to act as Authorized agent for our clients which helps reduce the time to import their products to India.
Registration of Medical Devices (Form 40, Form 41)
An application for issue of a Registration Certificate (Form 41) shall be made to the licensing authority in Form 40. The importer or the authorized agent in India having a valid whole sale license for sale or distribution of drugs can apply for the registration certificate in Form 40 on behalf of the manufacturer.
CDSCO has listed few medical devices under Notified category of medical devices. Only Notified Medical Devices must be registered at CDSCO, New Delhi.
Our dedicated team at CliniExperts can obtain Registration Certificate and subsequently Import License for our clients.
Re-registration of Medical Devices (Form 41)
Registration Certificate of Medical Devices has a 3 year validity. A minimum of 9 months before the expiry of this Registration Certificate, the importer has to file the application for Re-registration.
We assist our clients in the tracking of expiry of Registration Certificate and help them file the re-registration application before the given time enabling them hassle-free regulatory import compliance.
Import License for Medical Devices (Form 8, Form 10)
After receiving the registration certificate, an Import license must be obtained from CDSCO for import of medical equipments in India. We assist our clients infiling applications for import license expediting the process by filing right documents.
Amendments in Registration Certificate
Any change in the importer/manufacturer credentials must be informed to CDSCO. For this Amendment, a request must be made to the authorized officials so that the change can be incorporated in the Registration Certificate.
With years of experience and expertise in the field, we support our clients in filing the request to CDSCO,enabling successful Amendments in their Registration Certificate.
Marketing Authorization for Import New Medical Devices in India (Form 44 and 45)
New Medical Devices that are not approved in India require a Marketing Authorization from CDSCO to facilitate their import. It is a permission for the import of New Devices in India.
We assist our clients in obtaining the required Marketing Authorization from CDSCO without the lengthy paperwork and legwork.
For Manufacturer
Application for the grant of license for manufacture of notified or non-notified devices must be made to the State Licensing Authority (SLA) or Central Licenses Approving Authority (CLAA) depending upon product categorization, accompanied by the requisite fee in the manner prescribed in the Rules along with a copy to the office of DCG(I).
manufacturing license For Medical Devices From CLAA/SLA





- Permission to Manufacture
New Medical Devices in India
(Form 29, Form 44 and Form 46) - Manufacturing License for
Notified Medical devices from CLAA
(Form 27, Form 28) - Manufacturing License for
Non-Notified Medical Devices
from SLA (Form 27, Form 28)
Permission to Manufacture New Medical Devices in India (Form 29, Form 44 and Form 46)
The permission to manufacture any new medical device in India consists of three main forms- Form 29, Form 44 and Form 46.
Form 29 is a license to manufacture devices for the purpose of Examination Testing and Analysis and holds validity for one year.
Form 44 is an application for grant of permission to manufacture a New Medical Device.
Form 46 is Permission/Approval for manufacture of a new medical device.
We assist our clients in obtaining the permission successfully from the respective authorities enabling them to begin hassle free manufacturing.
Manufacturing License for Notified Medical devices from CLAA (Form 27, Form 28)
In India, application for the grant of licence for manufacturing Medical Devices is made in Form 27. It is for the grant or renewal of an existing licence to manufacture for sale or for distribution of devices specified in Schedules C and C (1). It is obtained from Central Licences Approving Authority (CLAA) in Form 28.
We provide end-to-end regulatory support to our clients in obtaining the required manufacturing license easily.
Manufacturing License for Non-Notified Medical Devices from SLA (Form 27, Form 28)
We can obtain manufacturing license for Non-Notified Medical Devices from SLA for our clients. We have presence in almost all states of India enabling us to effectively liaison with State FDA/ State Licensing Authority (SLA) for hassle free communication and license obtaining.
MDAC/SEC Meetings are important for categorizing medical devices in non-notified category as there is a fine line of difference. We also represent our clients in the MDAC/SEC meeting.
Clarification Regarding Medical Devices from CDSCO
The devicesthatare not listed under the Notified devices list as per the D&C Act, 1940 and Rules, 1945, require a clarification letter from CDSCO is required for its registration/clarification.
In some cases, for Class II and III devices, even if not classified in the notified devices category, the devices must be registered through this clarification letter.
We help our clients in preparing and submitting the letter for clarification to CDSCO.
Assistance on MDAC/SEC Meetings
The approval for device largely depends on the rationale and justification for using it. In line with the regulations at CDSCO, all cases of New Devices, Global Clinical Trials etc. are discussed by the MDAC/SEC committee of the respective therapeutic branch before the sponsor before granting an approval. CliniExperts have the capabilities to represent sponsor at the MDAC/SEC meeting. We can also evaluate sponsor’s proposal and can guide them to handle their cases at the at MDAC/SEC meeting.