Design , Packaging and labelling Solutions
End-to-End Solutions for Regulated Industries
We deliver regulatory-compliant design and packaging solutions tailored for Pharma, MedTech, Food, and Cosmetic brands in India
Compliance meets
Creativity
CliniExperts delivers an expanded suite of design, packaging,
and branding services, purpose-built for regulated industries
and fully aligned with Indian regulatory requirements.
Combined with our established regulatory and import expertise, we serve
as a single accountable partner for your compliance and creative needs.
Why Design and Packaging Need a Compliance
In India, packaging for regulated products must meet multiple specifications at once.
It should comply with labeling regulations, communicate information clearly, ensure safe use,
and still reflect your brand quality.
For pharma products, this means aligning with CDSCO requirements. For food and Nutraceuticals,
labels must follow FSSAI rules, including ingredients and claims. Medical devices require clear instructions, symbols, and safety information as per Medical Device Rules 2017, while cosmetics need proper ingredient listing, warnings, and compliant presentation as per Cosmetic Rules 2020.
That’s why we start every design with regulatory intent and not just aesthetics.
Our Capabilities
Sustainable Packaging
From concept to execution compliance-ready, market-aligned, and built for regulated sectors. Whether you are reviewing primary packaging concepts, secondary cartons, sleeves, labels, pouches, or rigid formats, we help create packaging that balances functionality, sustainability, and regulatory readiness.
Branding & Graphic Design
We develop brand and packaging artwork that reflects the seriousness of regulated sectors while remaining modern, clear, and market-ready.Cartons, labels, leaflets, pouches, tin cans, brochures, and catalogues crafted for pharma and food standards.
Digital Design
Today’s regulated brands also need a credible digital presence. We support this with digital design solutions that help companies present their products and capabilities with clarity and professionalism. Social media creatives, 3D mockups, and digital catalogues for a compelling modern brand presence.

Regulatory Compliance is the Foundation of our Work
At CliniExperts, regulatory compliance is not an added service layered onto creative work. It is the foundation of how we approach every deliverable. That means every design and packaging project is approached with a clear understanding of applicable framework, whether that involves CDSCO guidelines, the NDCT Rules, food labeling requirements, or other relevant Indian standards.
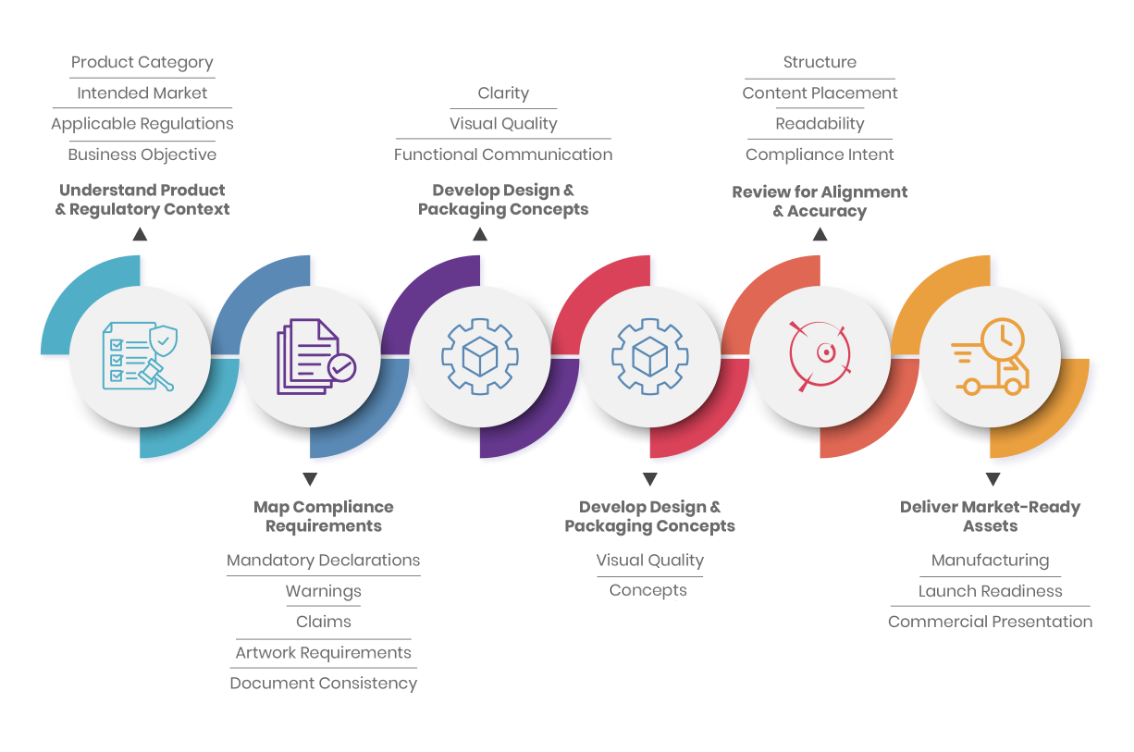
Our Approach
Compliance Driven Design Process
Industries we serve
India’s regulatory system extends beyond CDSCO, requiring approvals from multiple ministries. CliniExperts offers cross-functional expertise and continuous regulatory support.
FOOD

DRUGS & BIOLOGICAL

MEDICAL DEVICE
COSMETIC
Ready to take control of your regulatory ecosystem?
Connect with our regulatory strategists to build a tailored retainer program for your organization.
