Table of Contents
BIS Certification for imported products in India: Requirements & Process

BIS Certification of foreign manufacturers essential for ensuring that foreign products entering India meet safety, quality, and regulatory standards defined by the Bureau of Indian Standards.
Understanding BIS Certification for Importers
BIS Certification ensures that imported products comply with Indian safety and quality norms. It applies to electronics, appliances, and various regulated goods entering the Indian market.
Importers must ensure that foreign manufacturers obtain proper certification before selling products in India. This requirement helps maintain consumer safety and product reliability.
BIS License to import in India: Key Requirements
Obtaining a BIS license to import in India involves fulfilling several regulatory conditions. Importers can act as authorized representatives for foreign manufacturers.
Key requirements include:
- Valid business registration in India
- Authorization from foreign manufacturers
- Product testing in BIS-recognized labs
- Compliance with applicable Indian Standards
- Complete documentation submission
These requirements ensure accountability and traceability of imported products.
Foreign Product BIS Approval: Step-by-Step Process
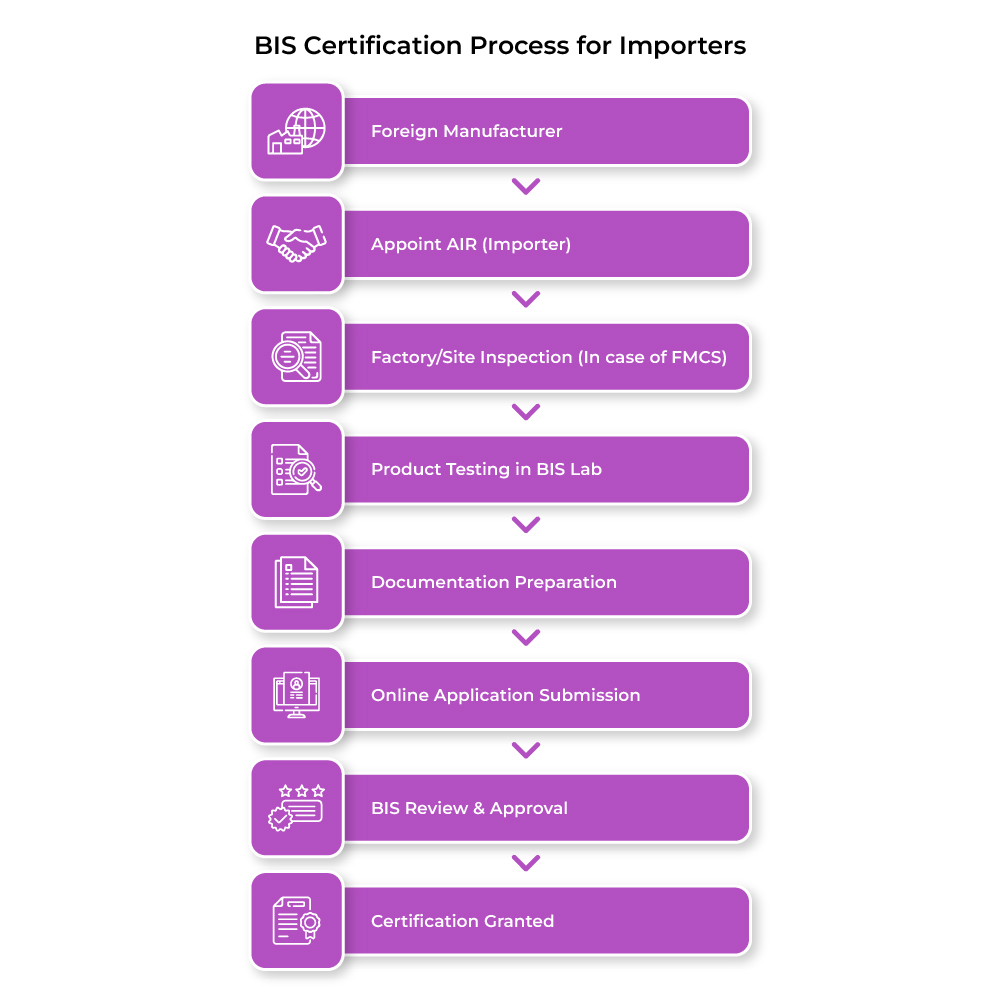
The BIS approval process for foreign products follows a structured framework similar to domestic certification procedures which is known as Foreign manufacturer certification scheme (FMCS). However, for products that fall under the Compulsory Registration Scheme (CRS), foreign manufacturers also have the option to obtain registration through the CRS route
1. Identification of Applicable Standards
Importers must identify relevant Indian Standards (IS codes) applicable to the product category before initiating the certification process.
2. Nomination of Authorized Indian Representative (AIR)
Foreign manufacturers must appoint an AIR in India. The importer often assumes this role for compliance and communication with BIS.
Audit/Inspection in case of foreign manufacturer certification scheme (FMCS):
BIS officer schedules a factory visits on a mutually agreed date and will collect samples and sent to an independent lab for testing.
3. Product Testing in BIS Labs
Products are tested in BIS-approved laboratories to ensure compliance with safety and quality standards.
4. Application Submission
Applications are submitted through the BIS online portal along with test reports and required documentation.
5. Grant of BIS Certification
After verification, BIS issues certification, allowing the importer to sell the product legally in India.
Table 1: Documents Required for BIS Certification for Importers
| Document Type | Description |
| Test Reports | From BIS-approved laboratories |
| Business Registration | Importer’s legal entity proof Manufacturer Factory/Company registration Manufacturer registered trademark |
| Authorization Letter | From foreign manufacturer |
| Product Details | Specifications and technical information |
| Application Forms | Completed BIS forms |
Challenges in BIS Certification for Importers
Importers often face challenges during certification due to complex regulations and procedural requirements.
Common issues include:
- Incomplete or incorrect documentation
- Delays in testing and reporting
- Misunderstanding of applicable standards
- Coordination issues with foreign manufacturers
Proper planning and expert guidance can help overcome these challenges efficiently.
Best Practices for Smooth BIS Import License India Process
Following best practices can simplify the certification process and reduce delays.
- Verify product category and standards early
- Maintain clear communication with manufacturers
- Ensure accurate documentation
- Choose reliable BIS-approved labs
- Monitor application status regularly
These steps help streamline the BIS import license India process effectively.
Role of Compliance in Foreign Product BIS Approval
Compliance plays a critical role in ensuring successful certification. It ensures that products meet Indian regulatory requirements and safety norms.
Adhering to BIS standards enhances product credibility and builds consumer trust in imported goods.
Conclusion
- BIS certification ensures safety and quality of imported products
- Importers act as key regulatory representatives
- Proper documentation and testing are essential
- Structured processes improve approval success
- Compliance enhances market credibility
In conclusion, BIS certification for importers is a mandatory and structured process that ensures foreign products meet Indian standards, enabling safe market entry and regulatory compliance.
Recent Posts
India vs ASEAN vs GCC: Choosing the Right Expansion Market for Pharmaceutical Companies

For global Pharmaceutical companies evaluating emerging market expansion, three regions consistently dominate strategic discussions: India, ASEAN, and the GCC. Each offers compelling growth characteri..
80 Categories, One Product: How to Get Cosmetic Product Categorisation Right in India

The Categorisation Problem Of all the challenges in India’s Cosmetic Import registration process, product categorisation is one of the most underestimated. Under the Cosmetics Rules, 2020, CDSCO..
Authorized Agent for Medical Device Import in India: When You Need One and How to Choose the Right Partner

For foreign Medical Device companies entering India, the authorized agent is not just a compliance checkpoint — it is a strategic regulatory role. The right appointment can support licensing procedu..
Need Help?
Submit your EnquiryOffice Locations
India
- Delhi
- Bangalore
Global
- USA
- Singapore
Call us on
Sales: +91 7672005050
Reception: +91-11-45214546
Timings
9 am to 6 pm (Monday to Friday)


