Table of Contents
How to Apply for BIS Certification in India: Process, Documents & Timeline

Businesses planning to apply BIS certification India must understand the regulatory framework governed by the Bureau of Indian Standards. The BIS certification process ensures product quality, safety, and compliance with national standards before entering the Indian market.
Manufacturers and importers often need BIS approval for regulated products such as electronics, Medical Devices, construction materials, and consumer goods. Understanding documentation requirements, testing procedures, and submission of workflows is critical for faster approval.
Understanding the BIS Certification Process in India
The BIS certification process is designed to verify that products meet Indian safety and quality standards. Certification is mandatory for several regulated product categories under BIS schemes.
Products are evaluated through product testing, conformity assessment, documentation review, and factory inspection. This structured approach ensures manufacturers maintain consistent quality standards throughout production.
The certification is typically granted under schemes such as ISI Mark Certification, Compulsory Registration Scheme (CRS), and Foreign Manufacturer Certification Scheme (FMCS).
BIS Application Checklist and Required Documentation
Before submitting an application, companies should prepare complete BIS certification documentation to avoid regulatory delays. Proper documentation simplifies the evaluation process and supports faster review.
Key documents required include:
- Business registration certificates
- Manufacturing license or company incorporation documents
- Product technical specifications
- Test reports from BIS-recognized laboratories
- Factory layout and manufacturing process details
- Quality control procedures and product labels
Maintaining a structured BIS application checklist helps organizations ensure no critical documentation is missed during submission.
Testing and Conformity Assessment Requirements
Product testing is a core part of the testing and conformity assessment stage of the BIS certification process. Products must comply with relevant Indian Standards (IS) before certification approval.
Testing must be conducted in BIS-recognized laboratories to verify product safety, performance, and quality parameters.
Manufacturers may also undergo factory inspection where BIS officials verify production capability, quality management systems, and product traceability.
BIS Certification Process: Step-by-Step Overview
Organizations planning to apply BIS certification India should follow a structured regulatory workflow to ensure compliance.
Typical steps include:
- Identify applicable BIS standards for the product
- Prepare BIS certification documentation and technical files
- Conduct product testing in BIS-approved laboratories
- Submit regulatory application through the BIS portal
- Undergo conformity assessment and factory inspection
- Receive BIS certification upon successful evaluation
These steps help ensure the product meets all safety, technical, and regulatory requirements.
Key Stages of BIS Certification in India
| Stage | Description |
| Application Submission | Manufacturer submits BIS application and documentation |
| Product Testing | Product tested in BIS-approved laboratory |
| Factory Inspection | BIS officials assess manufacturing facility |
| Conformity Assessment | Review of quality control and compliance |
| Certification Grant | BIS certification issued if requirements are satisfied |
BIS Certification Timeline and Approval Duration
The BIS certification timeline depends on product complexity, completeness of documentation, and testing requirements.
In general, the certification process may take several weeks to a few months depending on regulatory evaluation and inspection schedules.
Organizations can reduce approval delays by ensuring accurate documentation, timely laboratory testing, and proactive regulatory coordination.
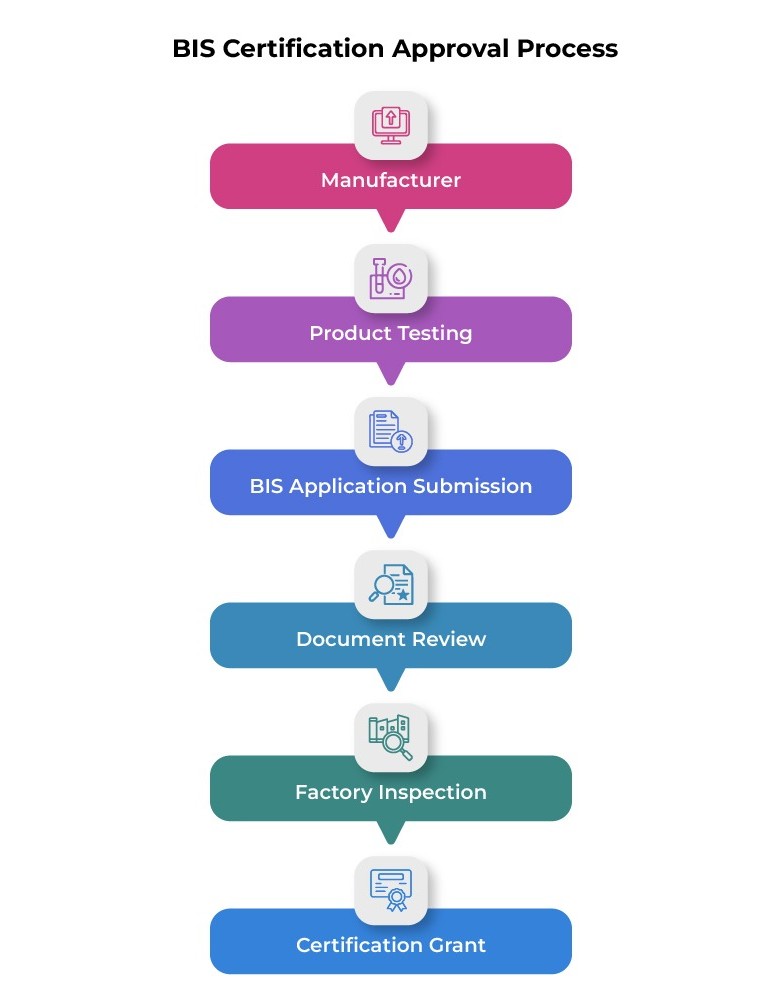
BIS Certification Approval Process
This diagram illustrates the regulatory workflow followed when companies apply for BIS certification, from testing and application submission to inspection and final certification approval.
Regulatory Submission India: Best Practices for Faster Approval
Preparing an accurate regulatory submission India package significantly improves approval timelines.
Organizations should focus on maintaining product documentation, ensuring compliance with applicable BIS standards, and conducting internal quality audits before submission.
Working with experienced regulatory consultants can also simplify the certification process and reduce the risk of application rejection.
Conclusion
Companies seeking market access must understand how to apply BIS certification India and comply with the regulatory requirements defined by the Bureau of Indian Standards. A clear understanding of documentation, testing procedures, and timelines helps organizations successfully navigate the BIS certification process.
Summary
- BIS certification ensures product safety and compliance with Indian standards
- Manufacturers must follow the structured BIS certification process
- Proper BIS certification documentation is critical for regulatory approval
- Testing and conformity assessment verify product quality and safety
- The BIS certification timeline varies depending on product category and inspection requirements
- Preparing a complete application helps organizations efficiently apply BIS certification India and obtain certification approval.
Saurangi is a food regulatory expert with 8 years of experience. She shares her knowledge and insights on regulatory updates, food trends, best practices, and news. Follow her for expert insights and practical advice on all things for food regulatory
Saurangi Shah
CliniExperts Services Pvt. Ltd.
Recent Posts
Documents Required for Cosmetic Import License in India (Checklist 2026)

The Central Drugs Standard Control Organization (CDSCO) is responsible for issuing Cosmetic Import licenses in India. Importers must apply through the SUGAM portal using Form COS-1 for fresh products...
Regulatory Due Diligence for Medical Devices in India: What Manufacturers Must Know Before Market Entry

Regulatory Due Diligence for Medical Devices in India is essential for manufacturers planning to enter the country’s rapidly growing healthcare market. Proper regulatory assessment ensures complianc..
Need Help?
Submit your EnquiryOffice Locations
India
- Delhi
- Bangalore
Global
- USA
- Singapore
Call us on
Sales: +91 7672005050
Reception: +91-11-45214546
Timings
9 am to 6 pm (Monday to Friday)


