Table of Contents
Regulatory Due Diligence for Medical Devices in India: What Manufacturers Must Know Before Market Entry

Regulatory Due Diligence for Medical Devices in India is essential for manufacturers planning to enter the country’s rapidly growing healthcare market. Proper regulatory assessment ensures compliance with CDSCO guidelines, licensing requirements, device classification rules, and safety standards.
India has strengthened its regulatory framework under the Medical Device Rules (MDR), 2017, making due diligence crucial before launching any Medical Device in the market.
Understanding Regulatory Due Diligence for Medical Devices in India
Regulatory Due Diligence for Medical Devices in India involves evaluating regulatory pathways, verifying compliance documentation, and ensuring devices meet safety and performance standards.
Manufacturers must analyze device classification, technical documentation, clinical evidence, and licensing obligations before submitting applications.
Conducting due diligence early reduces regulatory risks and accelerates market entry.
Medical Device Classification Under CDSCO
Medical Devices in India are categorized according to risk level. Regulatory authorities classify devices into four categories: Class A, B, C, and D.
The classification determines the regulatory pathway and approval requirements.
Table 1: Medical Device Risk Classification in India
| Device Class | Risk Level | Examples | Regulatory Authority |
| Class A | Low Risk | Cotton, surgical dressings | State Licensing Authority |
| Class B | Low-Moderate Risk | Thermometers | State Licensing Authority |
| Class C | Moderate-High Risk | Ventilators, implants | Central Licensing Authority (CDSCO) |
| Class D | High Risk | Heart valves | Central Licensing Authority (CDSCO) |
Licensing Requirements for Medical Device Market Entry
Manufacturers must obtain regulatory approval before marketing devices in India.
Key licensing requirements include:
- Import license for foreign manufacturers
- Manufacturing license for domestic producers
- Registration on the SUGAM MD Online Portal
- Compliance with Medical Device Rules (MDR), 2017
- Appointment of an Authorized Indian Agent for foreign manufacturers
Foreign companies must work with an authorized agent in India to submit regulatory applications and manage approvals.
Documentation Needed for Regulatory Approval
Proper documentation forms the backbone of regulatory compliance. Authorities evaluate device safety, performance, and manufacturing quality through submitted documents.
Important regulatory documents include:
- Power of Attorney (POA)
- Declaration of Conformity
- Free Sale Certificate (FSC)
- Quality Management System certification (ISO 13485)
- Plant Master File (PMF)
- Device Master File (DMF)
- Clinical evaluation reports
- Risk management documentation
- Verification and Validation
- Biocompatibility
- Post-Market Data
Comprehensive documentation improves approval timelines and regulatory success.
Regulatory Approval Process for Medical Devices in India
Manufacturers must follow a structured process before launching medical devices.
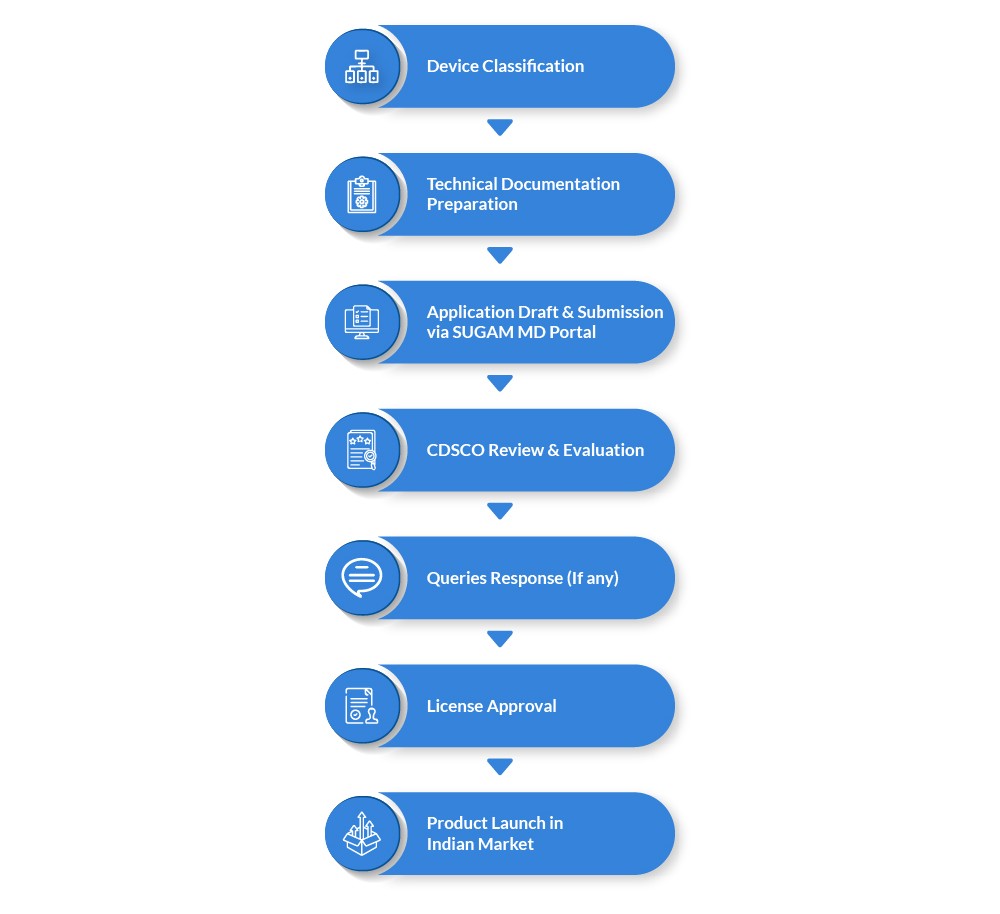
Common Regulatory Challenges for Manufacturers
Manufacturers entering the Indian market often face regulatory complexities.
Common challenges include:
- Incorrect device classification
- Incomplete documentation
- Delays in regulatory review
- Lack of clinical data
- Changing regulatory policies
Proper due diligence helps companies address these challenges effectively.
Role of Regulatory Consultants in India
Many manufacturers partner with regulatory consultants to navigate complex approval processes.
Regulatory experts assist with:
- CDSCO submission preparation
- Device classification assessment
- Regulatory documentation review
- Licensing and compliance strategies
Consulting firms provide end-to-end regulatory support for Medical Devices, Clinical Trials, and approvals across global markets.
Conclusion
Entering the Indian medical device market requires careful compliance planning and regulatory preparation. Regulatory Due Diligence for Medical Devices in India enables manufacturers to identify regulatory pathways, fulfill CDSCO requirements, and ensure product safety.
By performing thorough documentation review, classification analysis, and licensing preparation, companies can reduce regulatory risks. Conducting comprehensive Regulatory Due Diligence for Medical Devices in India ultimately supports faster approvals and successful market entry.
Summary
- Regulatory due diligence ensures compliance with CDSCO and MDR 2017 regulations.
- Medical devices are classified into Class A, B, C, and D based on risk levels.
- Licensing is required for manufacturing, import, and sell or distribution of devices.
- Regulatory submissions must be completed through the SUGAM MD Portal.
- Key documents include DMF, PMF, FSC, and Clinical Evaluation reports.
- Foreign manufacturers must appoint an authorized Indian agent for Importing the Product
Regulatory consultants can help simplify the approval process and reduce delays.
Saurangi is a food regulatory expert with 8 years of experience. She shares her knowledge and insights on regulatory updates, food trends, best practices, and news. Follow her for expert insights and practical advice on all things for food regulatory
Saurangi Shah
CliniExperts Services Pvt. Ltd.
Recent Posts
BIS Certification for imported products in India: Requirements & Process

BIS Certification of foreign manufacturers essential for ensuring that foreign products entering India meet safety, quality, and regulatory standards defined by the Bureau of Indian Standards. Underst..
Role & Responsibilities of Authorized Agent for Drugs in India

The role of authorized agent responsibilities is critical for foreign Pharmaceutical companies seeking to register, Import, and market Drugs in India while ensuring full regulatory compliance wi..
How to Get a CDSCO Import License in India: A Step-by-Step Guide for Foreign Medical Device Companies

In India given its large population and ample growth opportunities, remains one of the most attractive markets for global Medical Devices company. But entering Indian market is not so easy as it not o..
Need Help?
Submit your EnquiryOffice Locations
India
- Delhi
- Bangalore
Global
- USA
- Singapore
Call us on
Sales: +91 7672005050
Reception: +91-11-45214546
Timings
9 am to 6 pm (Monday to Friday)


