Regulation of 4 Medical Devices as Drugs by CDSCO: A new Notification
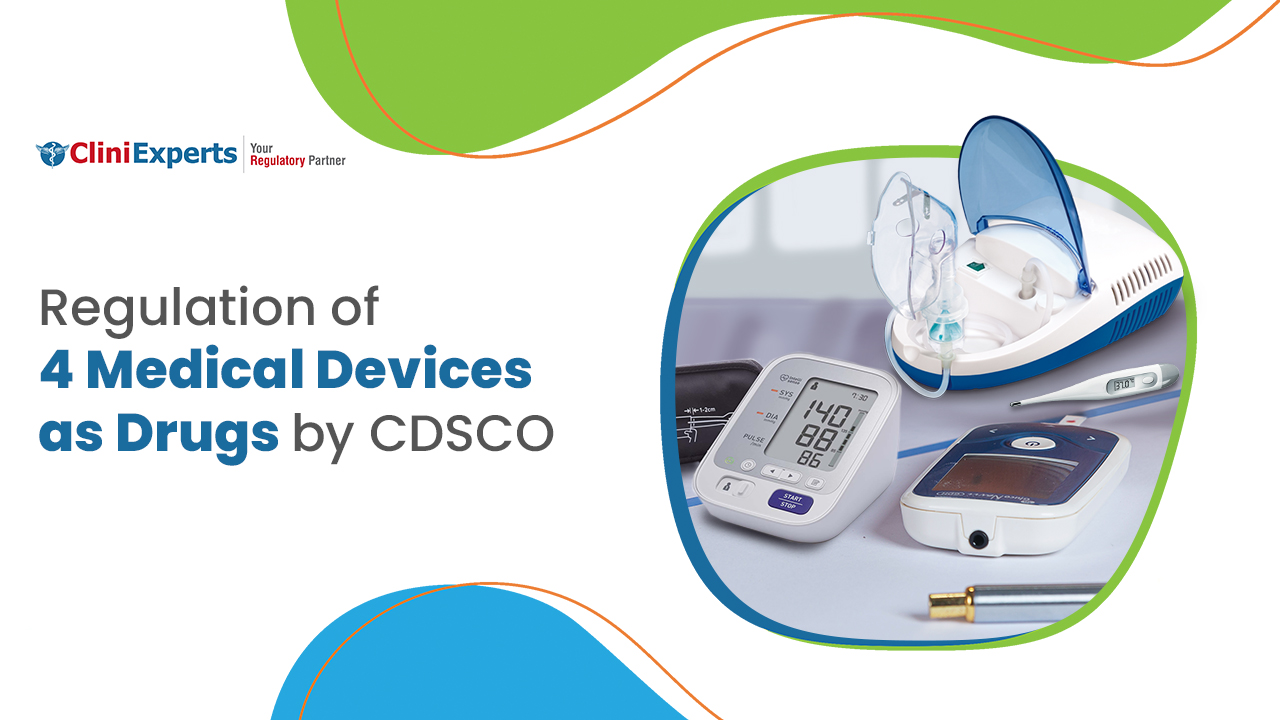
CDSCO has notified four medical devices, i.e., nebulizer, blood pressure monitoring devices, digital thermometer, and glucometer as drugs. The importers and manufacturers of these 4 medical devices need to take import and manufacturing licenses from the Central Licensing Authority or State Licensing Authority effectively from 1st January, 2021. According to Rule 97 of Medical Device Rules (MDR) 2017 and under the Drugs and Cosmetic Rules for the substances and devices referred to in rule 2 of the MDR, 2017.
Regarding the new notification, stakeholders of medical device industry approached the central authority to extend the implementation of 4 medical devices as drugs for another three to six months. There are lot of processes involved in the implementation of the notification like resolution of queries, audit of facilities by the regulatory authority and notified bodies in the country, and also testing of the products at the testing labs designated by the central authority. The circumstances where an existing importer or manufacturer is already importing or manufacturing any of these devices and submitted an application to the Central Licensing Authority or State Licensing Authority for import or manufacturing license for these medical devices under MDR,2017, the application will be considered valid. The importer can import the concerned medical device up to 6 months from the issuance of the order or till the Central licensing Authority or State Licensing Authority decide on the matter.
References:
- Regulation of Blood Glucose monitors Blood pressure monitors Nebulizers and Thermometers as drugs with effect from Jan 1st 2021. Available at: https://cdsco.gov.in/opencms/opencms/system/modules/CDSCO.WEB/elements/download_file_division.jsp?num_id=Njc5Mg== .Accessed on 14th February, 2021
Recent Posts
“The BIS–CDSCO Connection: Why Ninth Schedule Compliance Can Make or Break Your Cosmetic Import Registration in India”

Two Bodies, One Compliance Decision Most global Cosmetic brands entering India are familiar with CDSCO as the gatekeeper for Import registration. Far fewer understand the parallel role of the Bureau o..
MD-14 and MD-26 Explained: Import License Documents, Process, and Timelines for India

For any Foreign Medical Device company seeking to sell its devices in India, obtaining an Import License from CDSCO is a mandatory Regulatory requirement. The two central application forms in this pro..
CAR-T and Cell & Gene Therapy Regulation in India: What Global Developers Must Know

India’s Emerging Advanced Therapy Landscape Cell and Gene therapies — and chimeric antigen receptor T-cell (CAR-T) therapies in particular — represent one of the most transformative frontier..
Need Help?
Submit your EnquiryOffice Locations
India
- Delhi
- Bangalore
Global
- USA
- Singapore
Call us on
Sales: +91 7672005050
Reception: +91-11-45214546
Timings
9 am to 6 pm (Monday to Friday)


