
Blog
February 21, 2023

To import IVD devices in the indian market, it is important for importers to comprises with all the quality standards and competencies. The distribution, import, and manufacture of cosmetics, drugs, medical devices...

Blog
December 16, 2022

As per the rules put down by the Drugs and Cosmetics Rules, a wholesaler who wishes to sell, exhibit, store, or distribute drugs in India must have a wholesale drug license. The...

Blog
July 21, 2022

Class B Medical Devices / In-Vitro Diagnostic Kits Class B medical devices have low to moderate risk to the patients and public health risks. Class B medical devices’ permissions or approvals are...

Blog
July 7, 2022

Manufacturing And Product Development Support for Medical Devices / In-Vitro Diagnostic Kit The medical device industry of India is a crucial and integral part of the Indian healthcare industry. It provides preventative,...

Blog
June 20, 2022

Class A Medical Devices / IVDs – Class A medical devices / IVDs are medical equipment that has low risk to the patients and public health risks. Class A medical devices /...

Blog
June 16, 2022
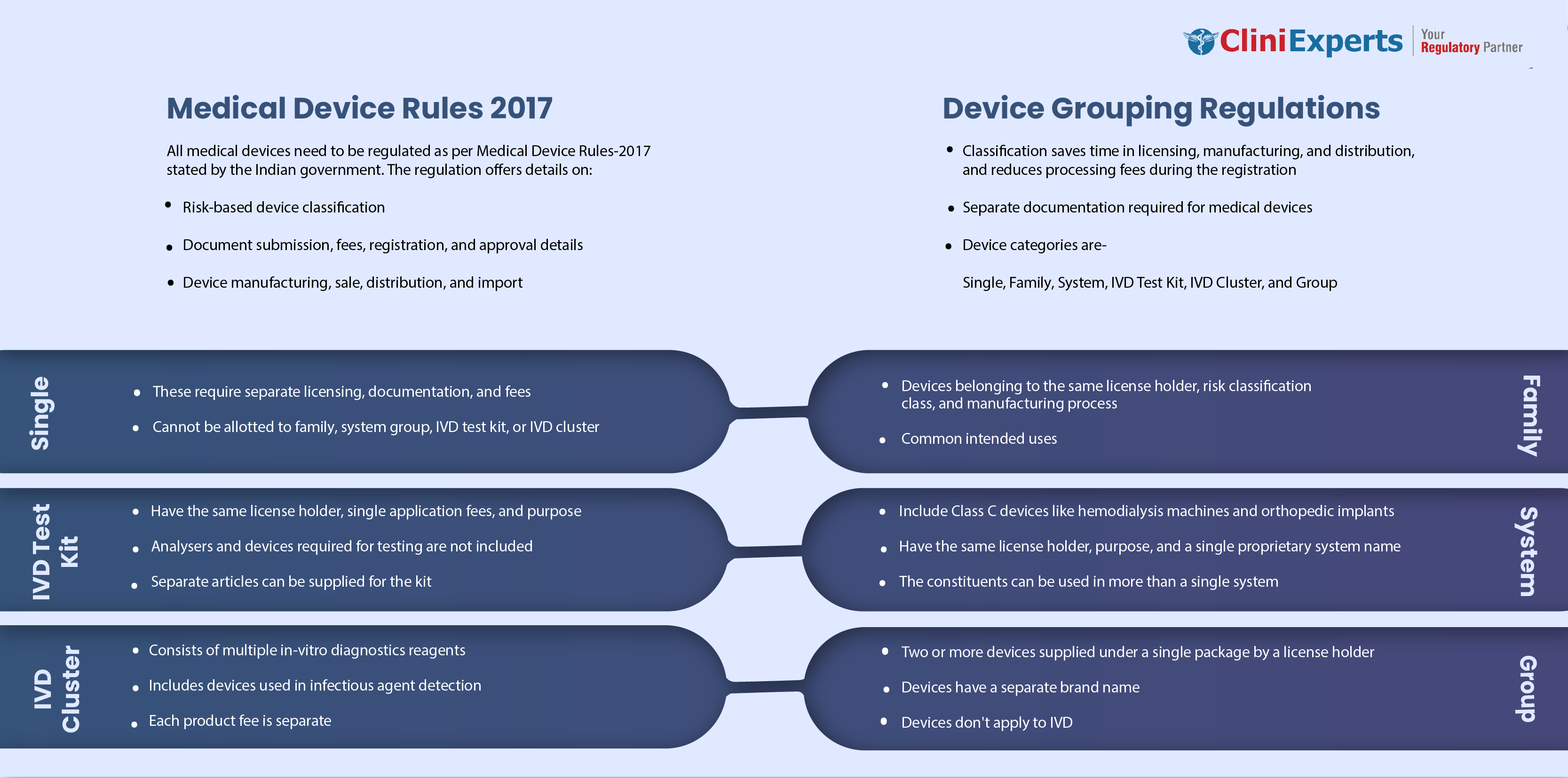
In the year 2017, the government of India announced that all the medical devices in India would be regulated as per Medical Device Rules-2017 (MDR-17), which gives a clear idea about the...

Regulatory Update
February 28, 2022

Regulatory Update
December 10, 2021







