Strategic Regulatory Outsourcing for Life Sciences
Scalable, Compliant, and Proven
With 15+ years of experience in India’s evolving regulatory landscape, we support pharma, medtech, and nutraceutical companies across every phase of the product lifecycle4through precision-driven, scalable outsourcing models.
To know more about the rules,
Please Fill the Form
To learn more, connect with us
Regulatory Outsourcing - A Strategic Imperative
In an industry where compliance directly impacts time-to-market, outsourcing is no longer a cost-cutting exercise- it’s a risk-mitigation and scalability strategy.

Maintain submission timelines across multiple jurisdictions

Manage region-specific formats like eCTD, XEVMPD, or India’s SUGAM Portal

Respond rapidly to agency queries and variations

Scale their regulatory capacity without bloating internal teams
We offer a modular regulatory outsourcing approach4enabling organizations to flex between specialized task support and full RA department outsourcing, depending on their internal maturity, project complexity, and market priorities.
Our Regulatory Outsourcing Models
We structure our engagement to match your operational architecture. Whether you need support for India-specific filings, global product launches, or ongoing compliance, our models ensure seamless integration with your internal teams and processes.
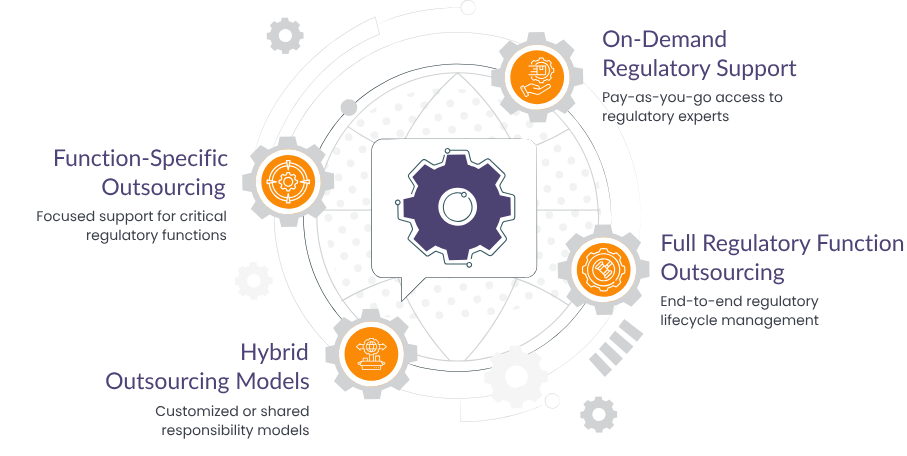
Function-Specific Outsourcing (FSP Model)
Focused support for critical regulatory functions
This model supports companies with in-house regulatory teams that need to offload repeatable, high-effort tasks- without losing ownership of regulatory strategy.
Use Cases
- Lifecycle document updates (e.g., labeling, SmPC, patient leaflets)
- Compilation and publishing in eCTD, NeeS, or country-specific formats
- Regulatory intelligence and health authority surveillance
- Clinical trial document handling and archiving (TMF compliance)
Expert Perspective
Many of our clients use this model to bridge operational gaps during pipeline expansion, product acquisition, or restructuring. Our teams function as an extension of your regulatory operations group, following your SOPs and quality systems.
On-Demand Regulatory Support (RAaaS)
Pay-as-you-go access to regulatory experts and infrastructure
This flexible model is ideal for dynamic regulatory environments, where unplanned variations, agency queries, or submissions arise at short notice.

Emergency Regulatory Response
Post-inspection commitments

Labeling Updates
Re-registrations in India

Gap Analysis
For regulatory submissions

Short-term Coverage
For staffing transitions or surges
Expert Perspective: Clients particularly value this model for its scalability and speed4we can deploy a regulatory publishing team within 24348 hours. Ideal for startups and SMEs who can’t afford full-time RA headcount but still need experienced resources on demand.

Full Regulatory Function Outsourcing
We manage your entire regulatory lifecycle - end to end
This model entrusts us with complete regulatory responsibility - from strategy through post-market compliance. We operate as your dedicated RA department, either globally or for specific markets like India.

India market entry for global manufacturers

NDA/BLA support from IND/CTA to post-approval changes

CDSCO representation and license holding (as Authorized Agent)

Health authority engagement and lifecycle management
Expert Perspective: For foreign companies without local presence, we act as the regulatory legal entity in India, holding import/manufacturer licenses and managing end-to-end submissions. We align timelines with your global strategy, avoiding duplication of efforts and ensuring compliance at every phase.
Hybrid Outsourcing Models
Customized or Shared Responsibility Models
In this collaborative model, clients retain high-level regulatory planning and decision-making, while outsourcing regional execution, operational activities, or specialized compliance tasks to us. A hybrid model combines elements of both in-house and outsourced regulatory functions. Instead of an “all or nothing” approach, companies retain certain regulatory responsibilities internally while outsourcing others.

Use Cases
- Internal strategy team manages global plan; we execute India filings
- Client manages pre-submission activities; we handle post-approval variations
- Shared responsibility for multi-region portfolio with centralized reporting

Expert Perspective
This model is often adopted during transitional phases 4such as mergers, rapid portfolio expansion, or geographic diversification. Our regulatory professionals integrate into your workflows, ensuring real-time communication and synchronized output.
Expertise Across Product Categories
Our deep regulatory knowledge spans across product types regulated under Indian and global frameworks:

Pharmaceuticals
APIs, generic drugs, NDAs, OTC formulations
Biologics
Biosimilars, cell and gene therapies, vaccines

Combination Products
Drug-device, biologic-device hybrids

In-Vitro Diagnostics
High-risk assays, companion diagnostics

Medical Devices
Class A to D, including software as a medical device (SaMD)

Cosmetics
Skin care, oral care, imported and domestic products

Nutraceuticals
Foods for special medical purposes, dietary supplements
Expert Perspective: We understand the nuances of CDSCO’s requirements and the practical application of Schedule M, Schedule Y, and the MDR/IVDR frameworks. Our consultants help you avoid regulatory pitfalls, reduce agency pushback, and maintain approval timelines.
Why Leading Companies Trust Us
With 15+ years of delivering regulatory solutions across 30+ countries, we’ve become the trusted outsourcing partner for startups, global pharma, and everything in between.
100+
Regulatory Experts
Across functional areas
30+
Countries Served
Global regulatory expertise
15+
Years Experience
Proven track record

Regulatory experts across functional areas
Dedicated Regulatory Intelligence Unit to track changing compliance requirements

Proven success with agencies like CDSCO, DCGI, FSSAI, EMA, US FDA, and WHO

India Authorized Agent Support with in-house license-holding capabilities
Expert Perspective: Our mission isn’t just to “get it done”4it’s to get it done right, on time, and in alignment with both local regulations and your global development plan.

Certifications and Accreditations
With these certifications, we demonstrate our dedication to quality, safety, and customer satisfaction.

ISO 9001

ISO 27001

ISO 13485

Wholesale

Ready to Transform Your Regulatory Strategy?
With 15+ years of delivering regulatory solutions across 30+ countries, we’ve become the trusted outsourcing partner for startups, global pharma, and everything in between.


Consultation
Discuss your regulatory needs and challenges

Assessment
Evaluate the best outsourcing model for your situation

Implementation
Seamless integration with your existing processes

Optimization
Continuous improvement of regulatory operations
Take the first step toward optimizing your regulatory operations. Our team of experts is ready to help you navigate the complex regulatory landscape and accelerate your path to market.
HAVE A QUERY?
REACH US!INDIA OFFICE
CLINIEXPERTS SERVICES PVT. LTD.
CliniExperts May assist you for:
- Product label compliance
- Regulatory monitoring
- Expanding into new geographies
- Product launches
- Product life cycle management
