Table of Contents
Inappropriate Use of Artificial Intelligence in Pharmaceutical Manufacturing

Artificial intelligence in pharmaceutical manufacturing can help support quality control, decision-making, process monitoring, batch evaluation, and data management by analysing complex production data to improve efficiency and product consistency. However, its use also introduces risks such as poor data quality, privacy concerns, regulatory challenges, lack of transparency, and high implementation costs. These limitations require strict validation, strong governance systems, and continuous human oversight to ensure safe, compliant, and reliable manufacturing outcomes.
Artificial Intelligence in Pharmaceutical Manufacturing
Artificial Intelligence (AI) can be used in Pharmaceutical manufacturing to improve quality control, decision-making, and production monitoring. It operates in regulated environments requiring safety, accuracy, and compliance. AI can manage large datasets, identify patterns, and support product quality. It assists human processes rather than replacing them, especially where precision and consistency are required. These are some keyways in which AI is globally being used in Pharmaceutical manufacturing today:
- Quality control support where it detects defects, anomalies, deviations using image recognition and computer vision.
- Decision-making support analyses manufacturing data, identifies patterns, trends, variations.
- Process monitoring tracks real-time production parameters, maintains controlled conditions.
- Batch evaluation assesses historical and real-time data, estimates quality outcomes.
- Data management organizes large manufacturing datasets, improves clarity and usability.1
While AI improves efficiency and quality control in Pharmaceutical manufacturing, it’s outputs must be carefully reviewed by an experienced human authority to ensure safety and compliance.
Risks and Challenges of AI in Pharmaceutical Manufacturing
Despite its advantages, the use of AI in pharmaceutical manufacturing presents several important risks and challenges. These risks are significant because pharmaceutical systems require high accuracy, validated processes, and strict compliance to ensure safe production outcomes. Any weakness in these areas can affect reliability, transparency, and overall system performance in real manufacturing environments.
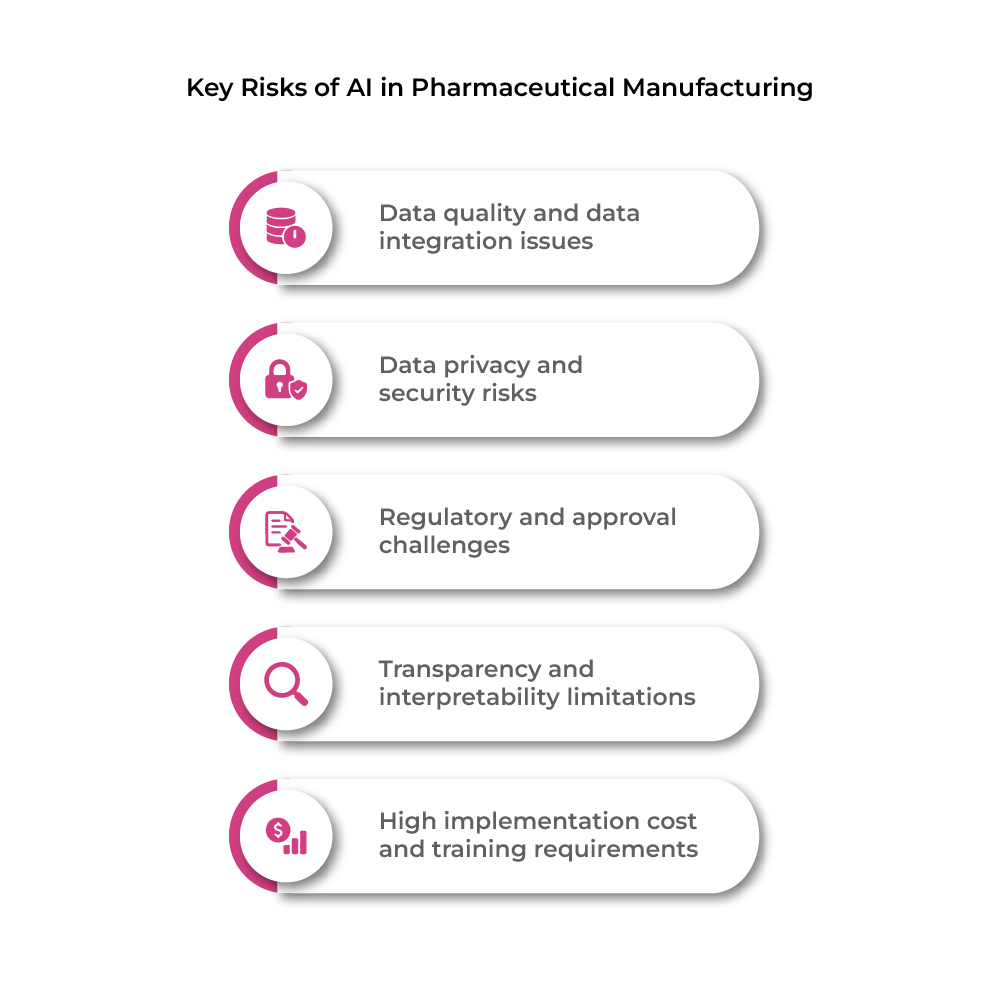
Table 1: Key Risks of AI in Pharmaceutical Manufacturing
| Risk Area | Description |
| Data quality and data integration issues | AI depends on large, structured datasets, but fragmented or inconsistent data reduces accuracy and limits analysis. |
| Data privacy and security risks | Sensitive patient and proprietary data may be exposed due to weak security controls, leading to regulatory violations. |
| Regulatory and approval challenges | Strict validation and regulatory approval from agencies like FDA and EMA are required before implementation. |
| Transparency and interpretability limitations | Many AI systems operate as “black boxes,” making decision logic difficult to explain in regulated environments. |
| High implementation cost and training requirements | AI deployment requires high investment, technical expertise, and workforce training. |
These risks highlight the importance of strict validation, governance, and human oversight in the use of artificial intelligence in pharmaceutical manufacturing. The following case study illustrates how improper application of artificial intelligence can lead to serious regulatory non-compliance and quality system failures.
Case Study: Inappropriate Use of AI in Pharmaceutical Manufacturing
The FDA issued a warning letter to Purolea Cosmetics Lab after identifying serious violations of Good Manufacturing Practice (CGMP) requirements during drug manufacturing operations. The inspection revealed failures in quality control systems and inappropriate reliance on artificial intelligence for generating regulatory documents, leading to non-compliance with Pharmaceutical manufacturing standards.3
Case Findings
• The quality control unit failed to properly supervise manufacturing operations and did not ensure that products met required standards of identity, strength, quality, and purity, indicating weak quality system implementation.3,4
• Manufacturing procedures were not properly established, reviewed, or consistently followed during production activities, showing failure in maintaining controlled and approved written processes required for Pharmaceutical manufacturing.3–5
• Batch production records were not reviewed before product release, reflecting inadequate quality control oversight and failure to ensure compliance before distribution.3,4
• Artificial intelligence tools were used to generate specifications and manufacturing documents without proper verification or approval by the quality control unit, resulting in regulatory non-compliance and weak validation practices.3
The FDA emphasized that while Artificial intelligence can assist in documentation and manufacturing support, it cannot replace mandatory human oversight. All AI-generated outputs must be reviewed and approved by the quality control unit to ensure compliance with CGMP regulations, proper documentation, and validated manufacturing processes.3–5
Regulatory Control of Artificial Intelligence Use in Pharmaceutical Manufacturing
Regulators such as the FDA, EMA, MHRA, and ICH ensure AI in Pharmaceutical manufacturing is used safely by enforcing strict GMP rules focused on risk, validation, and control. Their approach focuses on maintaining product quality, patient safety, and system reliability by ensuring that AI tools are appropriately validated, controlled throughout their lifecycle, and used within clearly defined operational limits.
Key regulatory controls
- AI systems must follow a risk-based regulatory approach, with stricter control for high-impact manufacturing decisions.
- Models are often required to be validated and “locked”, with any updates managed through formal change control systems.
- Strong enforcement of data quality principles (data must be complete, accurate, traceable, consistent, and reliable across its full lifecycle) ensures AI is trained and operated on trustworthy information.
- Human oversight is mandatory, so AI supports decisions but cannot fully replace expert judgment in GMP environments.
- Regulators require explainability and transparency, so AI outputs can be understood, audited, and justified scientifically.6
Conclusion
Artificial intelligence in Pharmaceutical manufacturing needs strict control to prevent unsafe or inappropriate use in regulated production systems. Unvalidated outputs have high risks when a human review is missing, or data quality is poor, which can lead to incorrect manufacturing decisions. A case example shows regulatory failure when artificial intelligence was used without proper approval in documentation processes.
Short Summary
- Artificial intelligence is being increasingly used in Pharmaceutical manufacturing to support quality control, decision-making, process monitoring, batch evaluation, and data management.
- It operates in a highly regulated environment where safety, accuracy, and compliance are essential.
- Artificial intelligence in Pharmaceutical manufacturing must be used under strict control to avoid unsafe or inappropriate applications in such a regulated environment.
- Inappropriate use can occur when artificial intelligence outputs are not properly validated, leading to faulty manufacturing decisions and risk to quality.
- Lack of human oversight may allow artificial intelligence generated results or documents to be used without proper review or approval.
- Regulatory systems and Good Manufacturing Practice rules require Artificial Intelligence models and outputs to be validated, monitored, traceable, and formally approved before use in manufacturing decisions.
References
1. Gonesh C, Saha G, Lima N, et al. Artificial Intelligence in Pharmaceutical Manufacturing: Enhancing Quality Control and Decision Making. Published online August 31, 2023.
2. *Roshan Madhukar pawar, 2 Vandana Shirsath, 3 Samrudhhi Sahane, 4 Ishita Chikhalikar, 5 Dr Anil, Jadhav. A Review article on Impact of Artificial Intelligence (AI) in Pharmaceutical Development and Manufacturing. Accessed April 21, 2026. https://ijirt.org/publishedpaper/IJIRT175127_PAPER.pdf
3. Purolea Cosmetics Lab – 722591 – 04/02/2026 | FDA. Accessed April 21, 2026. https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/purolea-cosmetics-lab-722591-04022026
4. eCFR :: 21 CFR 211.22 — Responsibilities of quality control unit. Accessed April 21, 2026. https://www.ecfr.gov/current/title-21/chapter-I/subchapter-C/part-211/subpart-B/section-211.22
5. eCFR :: 21 CFR 211.100 — Written procedures; deviations. Accessed April 21, 2026. https://www.ecfr.gov/current/title-21/chapter-I/subchapter-C/part-211/subpart-F/section-211.1006. Niazi SK. Regulatory Perspectives for AI/ML Implementation in Pharmaceutical GMP Environments. Pharmaceuticals. 2025;18(6):901. doi:10.3390/ph18060901
Recent Posts
The New CDSCO Environment: What Global Pharmaceutical Companies Must Know in 2026

Introduction India’s Pharmaceutical regulatory landscape is undergoing its most significant transformation in decades. For global Pharmaceutical companies — whether entering the Indian market ..
The Complete Roadmap to Cosmetic Import Registration in India

Introduction: India’s Cosmetic Market and Why Registration Matters India’s Cosmetic and personal care market has grown into one of the most consequential in the Asia-Pacific region — val..
Medical Device Classification in India: Class A, B, C, and D Explained for Importers and Manufacturers

Medical device classification is one of the first decisions that shapes a successful regulatory strategy in India. Whether a company plans to manufacture locally in India or import into the Indian mar..
Need Help?
Submit your EnquiryOffice Locations
India
- Delhi
- Bangalore
Global
- USA
- Singapore
Call us on
Sales: +91 7672005050
Reception: +91-11-45214546
Timings
9 am to 6 pm (Monday to Friday)


